- Blog
- About
- Contact
- Heucosin Pigment
- Nau Casino Night
- K Pots Vancouver Wa
- Casino 2000 Luxembourg
- Slots Gratuits Casino
- Casino Music The Beat Goes On
- I Occasionally Poop Blood
- 4 Education Lenses
- Slots Online Sites
- Ocasio Cortez 9/11 Quote
- Slots Earn Real Money
- Quotes Henry V
- Slots Games Win Money
- Casino 888 Free Blackjack
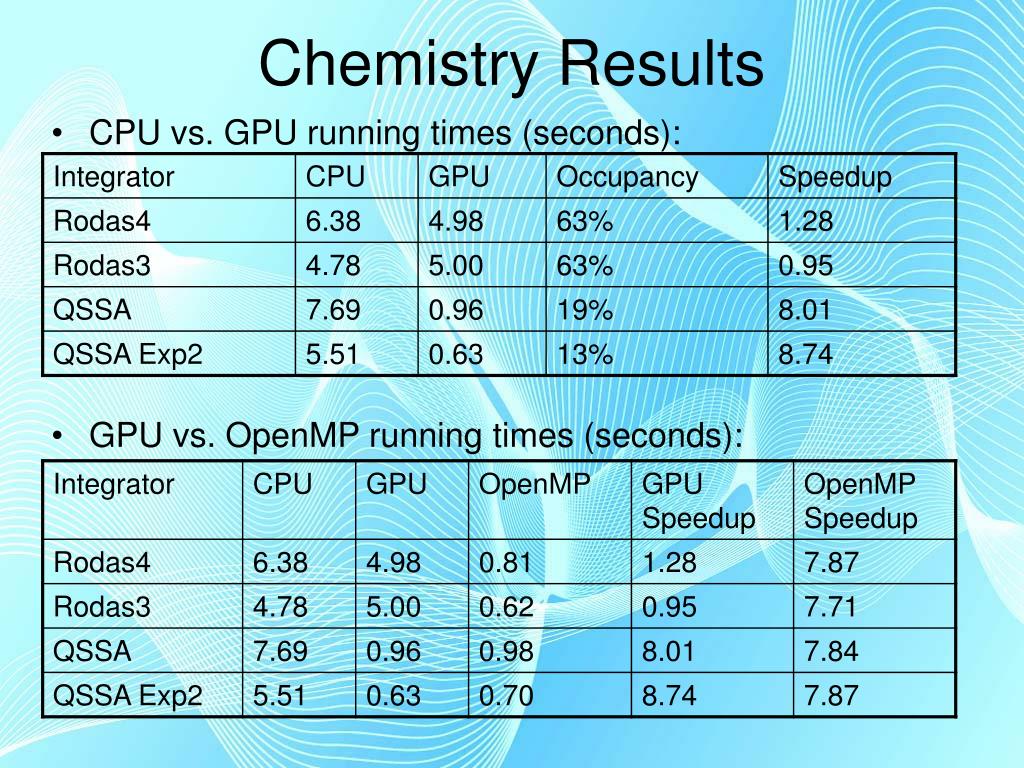
- Results In Chemistry
- Locational Boundary
- Planet 7 Casino Online Instant Play
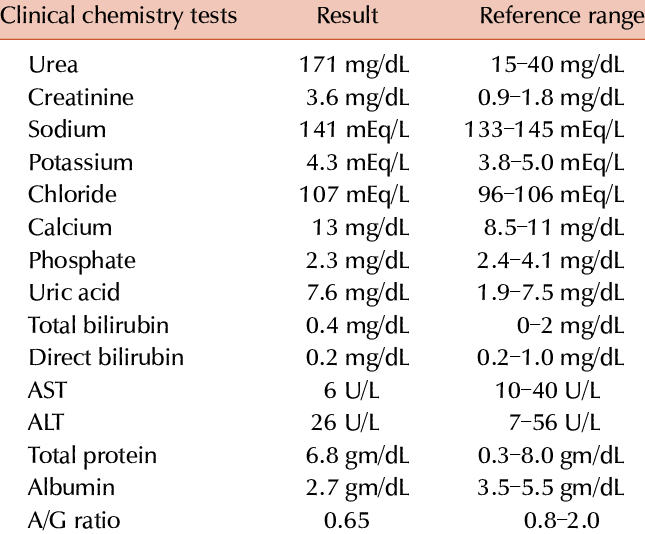
Chemistry (Chem) Screen
A large part of your laboratory report is results of the chemistry screen. These tests measure various chemicals in your blood to see whether your body is working correctly. See Fact Sheet 121 for information on the Complete Blood Count and Fact Sheet 123 for information on blood glucose (sugar) and cholesterol tests.
Comprehensive Metabolic Panel Results What Is a Comprehensive Metabolic Panel? Comprehensive metabolic panel (CMP) is a series of blood tests that give your doctor a snapshot of your body’s. Materials Chemistry: 2019: Q1: SJR The SJR is a size-independent prestige indicator that ranks journals by their 'average prestige per article'. It is based on the idea that 'all citations are not created equal'. SJR is a measure of scientific influence of journals that accounts for both the number of citations received by a journal and the. Writing a Lab Report (Chemistry) Underneath your tables/figures should be your written results. Write a paragraph or two to describe your data; explain what each figure means, and identify any trends. Be sure to be continuously refer back to the figures and tables as you explain your data (According to Table 2, etc.).
Laboratories have different 'reference ranges' or normal values for the results of each test. Most lab reports show the normal range and highlight any test results outside the normal range. For more information on normal laboratory test results, see Fact Sheet 120, Normal Laboratory Values.
The mineral calcium is a major component of bones and teeth. Calcium is also needed for nerves and muscles to work properly, and in chemical reactions in the cells. The body controls the amount of calcium in the blood. However, the amount of protein in the blood can affect calcium test results (see albumin). The most common cause of low calcium test results for people with HIV is low protein levels due to malnutrition or wasting. Abnormal calcium levels can indicate digestive problems.
Phosphorus, like calcium, is a major component of bones. Low levels of phosphorus for a long period of time can cause damage to bones, nerves and muscles. High phosphate levels are most often due to kidney failure.
Glucose is sugar, which is broken down in the cells to provide energy. See Fact Sheet 123 for more information on blood glucose tests.
The Electrolytes
Results In Chemistry Issn
The electrolytes are related to fluid balance in your cells. They are especially important if you become dehydrated or have kidney problems.
- Sodium levels indicate your balance of salt and water. They also are a sign of the functioning of your kidneys and adrenal glands. Abnormal blood sodium levels often indicate that blood volume is too low (due to dehydration) or too high. They can also occur when the heart is not pumping blood normally, or when the kidneys are not working properly.
- Potassium affects several major organs including the heart. Potassium levels rise in kidney failure, and may be abnormal due to vomiting or diarrhea.
- Chloride levels often go up and down along with sodium levels. This is because sodium chloride, or common salt, is a major component of blood.
- Bicarbonate or CO2 measures a buffer system in the blood. A normal CO2 level keeps the blood acidity at the correct level. A high level might be caused by high levels of lactic acid in the blood.
Kidney Function Tests
Bromine Test Results
The basic kidney function tests are blood urea nitrogen (BUN) and creatinine. Abnormal levels of phosphorus, sodium or uric acid can also be caused by kidney problems.
Blood Urea Nitrogen (BUN) is nitrogen in the blood. This is a waste product that is normally removed by the kidneys in the urine. High BUN levels can be due to a high-protein diet, dehydration, or kidney or heart failure.
Creatinine is a waste product of protein digestion and a measure of kidney function. High levels are usually due to kidney problems. Doctors use the creatinine level as the most direct sign of how well the kidneys are removing waste products from the body.
Liver Function Tests
The lab tests called 'liver function tests' actually measure the levels of enzymes found in the liver, heart, and muscles. Enzymes are proteins that cause or increase chemical reactions in living organisms. High enzyme levels can indicate liver damage caused by medications, alcohol, hepatitis, or recreational drug use.
Different patterns of these enzymes -- when some are elevated and others are normal -- can help your doctor identify specific health problems. Laboratory tests include:
- ALT (alanine aminotransferase), formerly called serum glutamate pyruvate transaminase or SGPT);used with the AST test to detect liver disease.
- AST (aspartate aminotransferase), formerly called serum glutamic-oxaloacetic transaminase or SGOT); used with the ALT test to detect liver disease.
- Bilirubin (a yellow fluid produced when red blood cells break down). High levels can indicate liver disease but might also be caused by the antiviral drugs indinavir (Crixivan) and atazanavir (Reyataz).
- Alkaline Phosphatase. High levels can indicate liver or bone disease.
- GGT (gamma glutamyl transpeptidase) results can show whether other abnormal test results are due to liver problems or bone problems.
- LDH (lactic dehydrogenase; not the same as lactic acid) is a general indicator of tissue damage.
Other Blood Chemistry Tests
Uric Acid comes from the breakdown of DNA (genetic material in the cells). It is normally removed by the kidneys. High levels of uric acid are fairly common. Very high levels can be caused when the kidneys are unable to remove uric acid from the blood or by leukemia or lymphoma.
Albumin is the major protein in the blood. It maintains water balance in the cells, carries nutrients to the cells and removes waste products. Low albumin is generally a sign of nutrition problems.
Because albumin carries so many substances in the blood, low albumin levels can cause incorrect low results for other laboratory tests, especially calcium or testosterone.
Globulin (also called immunoglobulin) measures the protein in antibodies produced by the immune system. HIV infection causes an abnormally high level of globulin. Levels are usually reported for five types of globulin: IgG, IgA, IgD, IgE and IgM.
Sedimentation Rate (Erythrocyte Sedimentation Rate) or Sed Rate measures how quickly red blood cells settle in a tube of blood. A high sed rate indicates some type of inflammation. However, the sed rate does not indicate whether the inflammation is long-term, like arthritis, or is due to the body fighting an infection.
The C-Reactive Protein test or CRP is another general test of inflammation. It rises and falls faster than the Sed Rate. High levels of CRP may be a sign of increased risk of heart attack.
For More Information
See Lab Tests Online at www.labtestsonline.org/ or go to MEDLINEplus at www.medlineplus.gov/ and search for a specific test.
Learning Objective
- Calculate the concentration of a diluted solution.
Key Points
- Most commonly, a solution’s concentration is expressed in terms of mass percent, mole fraction, molarity, molality, and normality. When calculating dilution factors, it is important that the units of volume and concentration remain consistent.
- Dilution calculations can be performed using the formula M1V1 = M2V2.
- A serial dilution is a series of stepwise dilutions, where the dilution factor is held constant at each step.
Terms
- dilutiona solution that has had additional solvent, such as water, added to make it less concentrated
- serial dilutionstepwise dilution of a substance in solution
Dilution refers to the process of adding additional solvent to a solution to decrease its concentration. This process keeps the amount of solute constant, but increases the total amount of solution, thereby decreasing its final concentration. Dilution can also be achieved by mixing a solution of higher concentration with an identical solution of lesser concentration. Diluting solutions is a necessary process in the laboratory, as stock solutions are often purchased and stored in very concentrated forms. For the solutions to be usable in the lab (for a titration, for instance), they must be accurately diluted to a known, lesser concentration.
The volume of solvent needed to prepare the desired concentration of a new, diluted solution can be calculated mathematically. The relationship is as follows:
[latex]M_1V_1=M_2V_2[/latex]
M1 denotes the concentration of the original solution, and V1 denotes the volume of the original solution; M2 represents the concentration of the diluted solution, and V2 represents the final volume of the diluted solution. When calculating dilution factors, it is important that the units for both volume and concentration are the same for both sides of the equation.
Impact Factor Of Chemistry Journals
Example
- 175 mL of a 1.6 M aqueous solution of LiCl is diluted with water to a final volume of 1.0 L. What is the final concentration of the diluted solution?
- [latex]M_1V_1=M_2V_2[/latex]
- (1.6 M)(175 mL) = M2(1000 mL)
- M2 = 0.28 M
Serial Dilutions
Serial dilutions involve diluting a stock or standard solution multiple times in a row. Typically, the dilution factor remains constant for each dilution, resulting in an exponential decrease in concentration. For example, a ten-fold serial dilution could result in the following concentrations: 1 M, 0.1 M, 0.01 M, 0.001 M, and so on. As is evidenced in this example, the concentration is reduced by a factor of ten in each step. Serial dilutions are used to accurately create extremely diluted solutions, as well as solutions for experiments that require a concentration curve with an exponential or logarithmic scale. Serial dilutions are widely used in experimental sciences, including biochemistry, pharmacology, microbiology, and physics.
Show SourcesBoundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources:
http://www.boundless.com/
Boundless Learning
CC BY-SA 3.0.
Boundless Learning
CC BY-SA 3.0.
http://en.wikipedia.org/wiki/serial%20dilution
Wikipedia
CC BY-SA 3.0.
Wikipedia
CC BY-SA 3.0.
http://en.wiktionary.org/wiki/dilution
Wiktionary
CC BY-SA 3.0.
Wiktionary
CC BY-SA 3.0.
http://en.wikipedia.org/wiki/Serial_dilution
Wikipedia
CC BY-SA 3.0.
Wikipedia
CC BY-SA 3.0.
“Sunil Kumar Singh, Dilution. September 17, 2013.”
http://cnx.org/content/m17123/latest/
OpenStax CNX
CC BY 3.0.
OpenStax CNX
CC BY 3.0.
http://commons.wikimedia.org/wiki/File:Dilution-concentration_simple_example.jpg
Wikimedia
CC BY-SA.
Wikimedia
CC BY-SA.
